

Name: Disodium Edetate
CAS No: Ethylenediaminetetraacetic acid, disodium salt [139-33-3] , Disodium ethylenediaminetetraacetate dihydrate [6381-92-6]
BP: Disodium Edetate JP: Disodium Edetate Hydrate PhEur: Disodium Edetate USP: Edetate Disodium
Dinatrii edetas; disodium EDTA; disodium ethylenediaminetetraacetate; edathamil disodium; edetate disodium; edetic acid, disodium salt.
Ethylenediaminetetraacetic acid, disodium salt [139-33-3] , Disodium ethylenediaminetetraacetate dihydrate [6381-92-6]
C10H14N2Na2O8 336.2 (for anhydrous) C10H18N2Na2O10 372.2 (for dihydrate
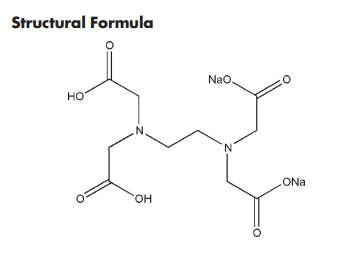
Disodium edetate is used as a chelating agent in a wide range of pharmaceutical preparations, including mouthwashes, ophthalmic preparations, and topical preparations,(1–3) typically at concentrations between 0.005 and 0.1% w/v. Disodium edetate forms stable water-soluble complexes (chelates) with alkaline earth and heavy-metal ions. The chelated form has few of the properties of the free ion, and for this reason chelating agents are often described as ‘removing’ ions from solution, a process known as sequestering. The stability of the metal–edetate complex is dependent on the metal ion involved and the pH. Disodium edetate is also used as a water softener as it will chelate calcium and magnesium ions present in hard water. It is also used therapeutically as an anticoagulant as it will chelate calcium and prevent the coagulation of blood in vitro. Concentrations of 0.1% w/v are used in small volumes for hematological testing and 0.3% w/v in transfusions.
Disodium edetate occurs as a white crystalline, odorless powder with a slightly acidic taste.
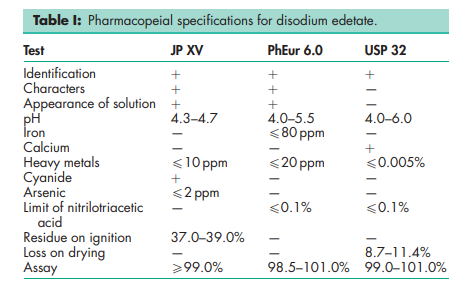
See Table I.
Acidity/alkalinity pH 4.3–4.7 (1% w/v solution in carbon dioxide-free water) Freezing point depression 0.148C (1% w/v aqueous solution) Melting point Decomposition at 2528C for the dihydrate. NIR spectra see Figure 1. Refractive index 1.33 (1% w/v aqueous solution) Solubility Practically insoluble in chloroform and ether; slightly soluble in ethanol (95%); soluble 1 part in 11 parts water. Specific gravity 1.004 (1% w/v aqueous solution) Viscosity (kinematic) 1.03 mm2 /s (1.03 cSt) (1% w/v aqueous solution).
Edetate salts are more stable than edetic acid (see also Edetic acid). However, disodium edetate dihydrate loses water of crystallization when heated to 1208C. Aqueous solutions of disodium edetate may be sterilized by autoclaving, and should be stored in an alkali-free container. Disodium edetate is hygroscopic and is unstable when exposed to moisture. It should be stored in a well-closed container in a cool, dry place.
Disodium edetate behaves as a weak acid, displacing carbon dioxide from carbonates and reacting with metals to form hydrogen. It is incompatible with strong oxidizing agents, strong bases, metal ions, and metal alloys.
Disodium edetate may be prepared by the reaction of edetic acid and sodium hydroxide.
Disodium edetate is used widely in topical, oral, and parenteral pharmaceutical formulations; it is used extensively in cosmetic and food products. Disodium edetate and edetate calcium disodium are used in a greater number and variety of pharmaceutical formulations than is edetic acid. Both disodium edetate and edetate calcium disodium are poorly absorbed from the gastrointestinal tract and are associated with few adverse effects when used as excipients in pharmaceutical formulations. Disodium edetate, trisodium edetate, and edetic acid readily chelate calcium and can, in large doses, cause calcium depletion (hypocalcemia) if used over an extended period of time, or if administered too rapidly by intravenous infusion. If used in preparations for the mouth, they can also leach calcium from the teeth. However, edetate calcium disodium does not chelate calcium. Disodium edetate should be used with caution in patients with renal impairment, tuberculosis, and impaired cardiac function. Although disodium edetate is generally considered safe, there have been reports of disodium edetate toxicity in patients receiving chelation therapy.(4) Nasal formulations containing benzalkonium chloride and disodium edetate, both known to be local irritants, were shown to produce an inflammatory reaction, and microscopic examination showed an extended infiltration of the mucosa by eosinophils, and pronounced atrophy and disorganization of the epithelium, although these effects were subsequently shown to be reversible.(3) The WHO has set an estimated acceptable daily intake for disodium EDTA in foodstuffs of up to 2.5 mg/kg body-weight.(5) See also Edetic acid. LD50 (mouse, IP): 0.26 g/kg(6) LD50 (mouse, IV): 0.056 g/kg LD50 (mouse, OP): 2.05 g/kg LD50 (rabbit, IV): 0.047 g/kg LD50 (rabbit, OP): 2.3 g/kg LD50 (rat, OP): 2.0 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Disodium edetate and its derivatives are mild irritants to the mucous membranes. Eye protection, gloves, and dust masks are recommended.
GRAS listed. Included in the FDA Inactive Ingredients Database (inhalations; injections; ophthalmic preparations; oral capsules, solutions, suspensions, syrups, and tablets; rectal topical, and vaginal preparations). Included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Edetic acid.
Disodium edetate has been used experimentally to investigate the stability and skin penetration capacity of captopril gel, in which disodium edetate was shown to exert a potent stabilizing effect, and may be used in the development of a transdermal drug delivery system.(7) A chitosan–EDTA conjugate has been investigated as a novel polymer for use in topical gels. The conjugate was shown to be stable, colorless, and transparent, and it also demonstrated antimicrobial effects.