

Name: Edetic Acid
CAS No: N,N-1,2-Ethanediylbis[N-(carboxymethyl)glycine] [60-00-4]
BP: Edetic Acid PhEur: Edetic Acid USP-NF: Edetic Acid
Acidum edeticum; Dissolvine; edathamil; EDTA; ethylenediaminetetraacetic acid; (ethylenedinitrilo)tetraacetic acid; Sequestrene AA; tetracemic acid; Versene Acid.
N,N-1,2-Ethanediylbis[N-(carboxymethyl)glycine] [60-00-4]
C10H16N2O8 292.24
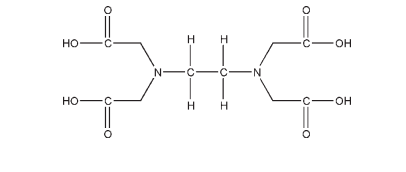
Edetic acid and edetate salts are used in pharmaceutical formulations, cosmetics, and foods as chelating agents. They form stable water-soluble complexes (chelates) with alkaline earth and heavy metal ions. The chelated form has few of the properties of the free ion, and for this reason chelating agents are often described as ‘removing’ ions from solution; this process is also called sequestering. The stability of the metal–edetate complex depends on the metal ion involved and also on the pH. The calcium chelate is relatively weak and will preferentially chelate heavy metals, such as iron, copper, and lead, with the release of calcium ions. For this reason, edetate calcium disodium is used therapeutically in cases of lead poisoning; see also Section 18. Edetic acid and edetates are primarily used as antioxidant synergists, sequestering trace amounts of metal ions, particularly copper, iron, and manganese, that might otherwise catalyze autoxidation reactions. Edetic acid and edetates may be used alone or in combination with true antioxidants, the usual concentration employed being in the range 0.005–0.1% w/v. Edetates have been used to stabilize ascorbic acid; corticosteroids; epinephrine; folic acid; formaldehyde; gums and resins; hyaluronidase; hydrogen peroxide; oxytetracycline; penicillin; salicylic acid, and unsaturated fatty acids. Essential oils may be washed with a 2% w/v solution of edetate to remove trace metal impurities. Edetic acid and edetates possess some antimicrobial activity but are most frequently used in combination with other antimicrobial preservatives owing to their synergistic effects. Many solutions used for the cleaning, storage, and wetting of contact lenses contain disodium edetate. Typically, edetic acid and edetates are used in concentrations of 0.01–0.1% w/v as antimicrobial preservative synergists; see Section 10. Edetic acid and disodium edetate may also be used as water softeners since they will chelate the calcium and magnesium ions present in hard water; edetate calcium disodium is not effective. Many cosmetic and toiletry products, e.g. soaps, contain edetic acid as a water softener.
Edetic acid occurs as a white crystalline powder.
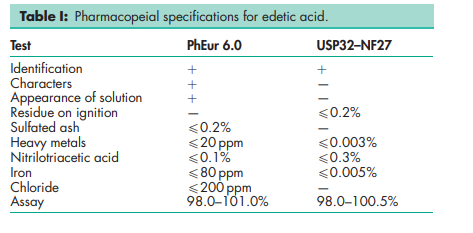
See Table I.
Acidity/alkalinity pH = 2.2 for a 0.2% w/v aqueous solution. Antimicrobial activity Edetic acid has some antimicrobial activity against Gram-negative microorganisms, Pseudomonas aeruginosa, some yeasts, and fungi although this activity is insufficient for edetic acid to be used effectively as an antimicrobial preservative on its own.(1,2) However, when used with other antimicrobial preservatives, edetic acid demonstrates a marked synergistic effect in its antimicrobial activity. Edetic acid and edetates are therefore frequently used in combination with such preservatives as benzalkonium chloride; bronopol; cetrimide; imidurea; parabens; and phenols, especially chloroxylenol. Typically, edetic acid is used at a concentration of 0.1–0.15% w/v. In the presence of some divalent metal ions, such as Ca2þ or Mg2þ, the synergistic effect may be reduced or lost altogether. The addition of disodium edetate to phenylmercuric nitrate(3) and thimerosal(3,4) has also been reported to reduce the antimicrobial efficacy of the preservative. Edetic acid and iodine form a colorless addition compound that is bactericidal. Dissociation constant pKa1 = 2.00; pKa2 = 2.67; pKa3 = 6.16; pKa4 = 10.26. Melting point Melts above 2208C, with decomposition. NIR spectra see Figure 1. Solubility Soluble in solutions of alkali hydroxides; soluble 1 in 500 of water.
Although edetic acid is fairly stable in the solid state, edetate salts are more stable than the free acid, which decarboxylates if heated above 1508C. Disodium edetate dihydrate loses water of crystallization when heated to 1208C. Edetate calcium disodium is slightly hygroscopic and should be protected from moisture. Aqueous solutions of edetic acid or edetate salts may be sterilized by autoclaving, and should be stored in an alkali-free container. Edetic acid and edetates should be stored in well-closed containers in a cool, dry place.
Edetic acid and edetates are incompatible with strong oxidizing agents, strong bases, and polyvalent metal ions such as copper, nickel, and copper alloy. Edetic acid and disodium edetate behave as weak acids, displacing carbon dioxide from carbonates and reacting with metals to form hydrogen. Other incompatibilities include the inactivation of certain types of insulin due to the chelation of zinc, and the chelation of trace metals in total parenteral nutrition (TPN) solutions following the addition of TPN additives stabilized with disodium edetate. Calcium disodium edetate has also been reported to be incompatible with amphotericin and with hydralazine hydrochloride in infusion fluids.
Edetic acid may be prepared by the condensation of ethylenediamine with sodium monochloroacetate in the presence of sodium carbonate. An aqueous solution of the reactants is heated to about 908C for 10 hours, then cooled, and hydrochloric acid is added to precipitate the edetic acid. Edetic acid may also be prepared by the reaction of ethylenediamine with hydrogen cyanide and formaldehyde with subsequent hydrolysis of the tetranitrile, or under alkaline conditions with continuous extraction of ammonia.
Edetic acid and edetates are widely used in topical, oral, and parenteral pharmaceutical formulations. They are also extensively used in cosmetics and food products. Edetic acid is generally regarded as an essentially nontoxic and nonirritant material, although it has been associated with doserelated bronchoconstriction when used as a preservative in nebulizer solutions. It has therefore been recommended that nebulizer solutions for bronchodilation should not contain edetic acid.(5) Edetates, particularly disodium edetate and edetate calcium disodium, are used in a greater number and variety of pharmaceutical formulations than the free acid. Disodium edetate, trisodium edetate, and edetic acid readily chelate calcium and can, in large doses, cause calcium depletion (hypocalcemia) if used over an extended period or if administered too rapidly by intravenous infusion. If used in preparations for the mouth, they can also leach calcium from the teeth. In contrast, edetate calcium disodium does not chelate calcium. Edetate calcium disodium is nephrotoxic and should be used with caution in patients with renal impairment. The WHO has set an estimated acceptable daily intake for disodium edetate in foodstuffs at up to 2.5 mg/kg body-weight.(6) LD50 (mouse, IP): 0.25 g/kg(7) LD50 (rat, IP): 0.397 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Edetic acid and edetates are mildly irritant to the skin, eyes, and mucous membranes. Ingestion, inhalation, and contact with the skin and eyes should therefore be avoided. Eye protection, gloves, and a dust mask are recommended
Included in the FDA Inactive Ingredients Database (oral, otic, rectal, and topical preparations; submucosal injection preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Dipotassium edetate; disodium edetate; edetate calcium disodium; sodium edetate; trisodium edetate. Dipotassium edetate Empirical formula C10H14K2N2O8 Molecular weight 368.46 CAS number [2001-94-7] Synonyms Dipotassium edathamil; dipotassium ethylenediaminetetraacetate; edathamil dipotassium; edetate dipotassium; edetic acid dipotassium salt; EDTA dipotassium; N,N0 -1,2-ethanediylbis[N-(carboxymethyl)glycine] dipotassium salt; ethylenebis (iminodiacetic acid) dipotassium salt; ethylenediaminetetraacetic acid dipotassium salt; (ethylenedinitrilo)tetraacetic acid dipotassium salt; tetracemate dipotassium. Appearance White crystalline powder. Comments The EINECS number for dipotassium edetate is 217- 895-0. Edetate calcium disodium Empirical formula C10H12CaN2Na2O8 Molecular weight 374.28 CAS number [62-33-9] for the anhydrous material and [23411- 34-9] for the dihydrate Synonyms Calcium disodium edetate; calcium disodium ethylenediaminetetraacetate; calcium disodium (ethylenedinitrilo) tetraacetate; E385; edathamil calcium disodium; edetic acid calcium disodium salt; EDTA calcium; ethylenediaminetetraacetic acid calcium disodium chelate; [(ethylenedinitrilo)tetraacetato]calciate(2-) disodium; sodium calciumedetate; Versene CA. Appearance White or creamy-white colored, slightly hygroscopic, crystalline powder or granules; odorless, or with a slight odor; tasteless, or with a faint saline taste. Acidity/alkalinity pH = 4–5 for a 1% w/v aqueous solution. Density (bulk) 0.69 g/cm3 Solubility Practically insoluble in chloroform, ether, and other organic solvents; very slightly soluble in ethanol (95%); soluble 1 in 2 of water. Method of manufacture Edetate calcium disodium may be prepared by the addition of calcium carbonate to a solution of disodium edetate. Safety see also Section 14. LD50 (mouse, IP): 4.5 g/kg(7) LD50 (rabbit, IP): 6 g/kg LD50 (rabbit, oral): 7 g/kg LD50 (rat, IP): 3.85 g/kg LD50 (rat, IV): 3.0 g/kg LD50 (rat, oral): 10 g/kg Regulatory status GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (injections; oral capsules, solutions, suspensions, syrups, and tablets). Comments Used in pharmaceutical formulations as a chelating agent in concentrations between 0.01–0.1% w/v. Usually edetate calcium disodium is used in pharmaceutical formulations in preference to disodium edetate or sodium edetate to prevent calcium depletion occurring in the body. In food products, edetate calcium disodium may also be used in flavors and as a color retention agent. Edetate calcium disodium occurs as the dihydrate, trihydrate, and anhydrous material. Some pharmacopeias specify that edetate calcium disodium is the dihydrate, others that it is the anhydrous material. The USP 32 specifies that edetate calcium disodium is a mixture of the dihydrate and trihydrate but that the dihydrate predominates. Edetate calcium disodium is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. The EINECS number for edetate calcium disodium is 200- 529-9. Sodium edetate Empirical formula C10H12N2Na4O8 Molecular weight 380.20 CAS number [64-02-8] Synonyms Edetate sodium; edetic acid tetrasodium salt; EDTA tetrasodium; N,N0 -1,2-ethanediylbis[N-(carboxymethyl)glycine] tetrasodium salt; ethylenebis(iminodiacetic acid) tetrasodium salt; ethylenediaminetetraacetic acid tetrasodium salt; (ethylenedinitrilo)tetraacetic acid tetrasodium salt; Sequestrene NA4; tetracemate tetrasodium; tetracemin; tetrasodium edetate; tetrasodium ethylenebis(iminodiacetate); tetrasodium ethylenediaminetetraacetate; Versene. Appearance White crystalline powder. Acidity/alkalinity pH = 11.3 for a 1% w/v aqueous solution. Melting point >3008C Solubility Soluble 1 in 1 of water. Safety see also Section 14. LD50 (mouse, IP): 0.33 g/kg(7) Regulatory status Included in the FDA Inactive Ingredients Database (inhalations; injections, ophthalmic preparations, oral capsules and tablets; and topical preparations). Comments Sodium edetate reacts with most divalent and trivalent metallic ions to form soluble metal chelates and is used in pharmaceutical formulations in concentrations between 0.01–0.1% w/v. Trisodium edetate Empirical formula C10H13N2Na3O8 Molecular weight 358.20 CAS number [150-38-9] Synonyms Edetate trisodium; edetic acid trisodium salt; EDTA trisodium; N,N0 -1,2-ethanediylbis[N-(carboxymethyl)glycine] trisodium salt; ethylenediaminetetraacetic acid trisodium salt; (ethylenedinitrilo)tetraacetic acid trisodium salt; Sequestrene NA3; trisodium ethylenediaminetetraacetate; Versene-9. Appearance White crystalline powder. Acidity/alkalinity pH = 9.3 for a 1% w/v aqueous solution. Melting point >3008C Method of manufacture Trisodium edetate may be prepared by adding a solution of sodium hydroxide to disodium edetate. Safety see also Section 14. LD50 (mouse, IP): 0.3 g/kg(7) LD50 (mouse, oral): 2.15 g/kg LD50 (rat, oral): 2.15 g/kg Regulatory status Included in the FDA Inactive Ingredients Database (topical preparations). Comments More soluble in water than either the disodium salt or the free acid. Trisodium edetate also occurs as the monohydrate and is used in pharmaceutical formulations as a chelating agent. The EINECS number for trisodium edetate is 205-758-8
Other salts of edetic acid that are commercially available include diammonium, dimagnesium, ferric sodium, and magnesium disodium edetates. Therapeutically, a dose of 50 mg/kg body-weight of disodium edetate, as a slow infusion over a 24-hour period, with a maximum daily dose of 3 g, has been used as a treatment for hypercalcemia. For the treatment of lead poisoning, a dose of 60–80 mg/kg of edetate calcium disodium, as a slow infusion in two daily doses, for 5 days, has been used. Chelation therapy using edetic acid has been widely used for the treatment of ischemic heart disease. However, it has been suggested that the therapeutic benefits of this treatment may be due to the changes in lifestyle of the patient rather than the administration of edetic acid (40 mg/kg by infusion over a 3-hour period).(8) The EINECS number for edetic acid is 200-449-4.