

Name: Erythorbic Acid
CAS No: Isoascorbic acid [89-65-6]
USP-NF: Erythorbic Acid
Araboascorbic acid; d-araboascorbic acid; D-2,3-didehydro-erythro-hexono-1,4-lactone; E315; erycorbin; d-erythorbic acid; Derythro-hex-2-enoic acid; D-erythro-3-ketohexonic acid lactone; glucosaccharonic acid; D-isoascorbic acid; isovitamin C; g-lactone; saccharosonic acid.
Isoascorbic acid [89-65-6]
C6H8O6 176.14
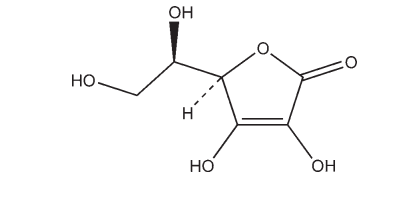
Erythorbic acid is a stereoisomer of L-ascorbic acid, and is used as an antioxidant in foods and oral pharmaceutical formulations. It has approximately 5% of the vitamin C activity of L-ascorbic acid.
Erythorbic acid occurs as a white or slightly yellow-colored crystals or powder. It gradually darkens in color upon exposure to light.
See Table I.

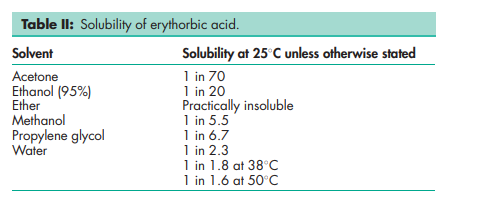
Acidity/alkalinity pH = 2.1 (10% w/v aqueous solution at 258C) Density (bulk) 0.704 g/cm3 Melting point 164–1718C with decomposition at 1848C Solubility see Table II. Specific rotation [a]D 20 = 16.5 to 18.08 (10% w/v aqueous solution)
Erythorbic acid should be stored in an airtight container, protected from light, in a cool, dry place.
Erythorbic acid is incompatible with chemically active metals such as aluminum, copper, magnesium, and zinc. It is also incompatible with strong bases and strong oxidizing agents.
Erythorbic acid is synthesized by the reaction between methyl 2- keto-D-gluconate and sodium methoxide. It can also be synthesized from sucrose, and produced from Penicillium spp
Erythorbic acid is widely used in food applications as an antioxidant. It is also used in oral pharmaceutical applications as an antioxidant. Erythorbic acid is generally regarded as nontoxic and nonirritant when used as an excipient. Erythorbic acid is readily metabolized and does not affect the urinary excretion of ascorbic acid. The WHO has set an acceptable daily intake of erythorbic acid and its sodium salt in foods at up to 5 mg/kg body-weight.(1)
Observe normal precautions appropriate to the circumstances and quantity of material handled. When heated to decomposition, erythorbic acid emits acrid smoke and irritating fumes
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (oral concentrate and tablets).
Ascorbic acid; sodium erythorbate Sodium erythorbate Empirical formula C6H7NaO6 Molecular weight 198.11 CAS number [7378-23-6] Synonyms E316; D-erythro-hex-2-enoic acid sodium salt; erythorbic acid sodium salt. Acidity/alkalinity pH = 7.2–7.9 for 10% w/v aqueous solution. Melting point 1728C Solubility Soluble 1 in 6.5 of water. The sodium salt is less soluble in water than the free acid. Comments The EINECS number for sodium erythorbate is 228- 973-6
A specification for erythorbic acid is included in the Food Chemicals Codex (FCC).(2) The EINECS number for erythorbic acid is 201-928-0. The PubChem Compound ID (CID) for erythorbic acid is 6981