

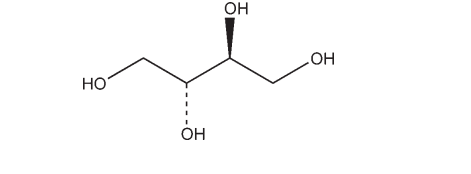
Name: Erythritol
CAS No: (2R,3S)-Butane 1,2,3,4-tetrol [149-32-6]
BP: Erythritol PhEur: Erythritol USP-NF: Erythritol
Butane 1,2,3,4-tetrol; 1,2,3,4-butanetetrol; C*Eridex; E968; erythrite; erythritolum; erythroglucin; meso-erythritol; phycite; tetrahydroxybutane; Zerose.
(2R,3S)-Butane 1,2,3,4-tetrol [149-32-6]
C4H10O4 122.12
Erythritol is a naturally occurring noncariogenic excipient used in a variety of pharmaceutical preparations, including in solid dosage forms as a tablet filler,(1) and in coatings.(2) It has also been investigated for use in dry powder inhalers.(3,4) It is also used in sugar-free lozenges,(5,6) and medicated chewing gum.(5 Erythritol can also be used as a diluent in wet granulation in combination with moisture-sensitive drugs.(7) In buccal applications, such as medicated chewing gums, it is used because of its high negative heat of solution which provides a strong cooling effect. Erythritol is also used as a noncaloric sweetener in syrups;(8) it is used to provide sensorial profile-modifying properties with intense sweeteners; and it is also used to mask unwanted aftertastes. Erythritol is also used as a noncariogenic sweetener in toothpastes and mouthwash solutions. See Table I.
Erythritol is a sugar alcohol (polyol) that occurs as a white or almost white powder or granular or crystalline substance. It is pleasant tasting with a mild sweetness approximately 60–70% that of sucrose. It also has a high negative heat of solution that provides a strong cooling effect.
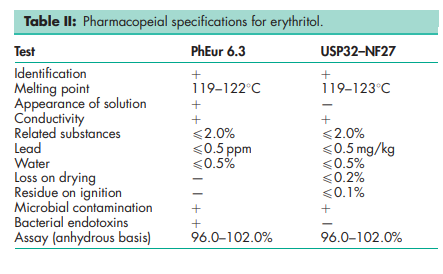
See Table II.
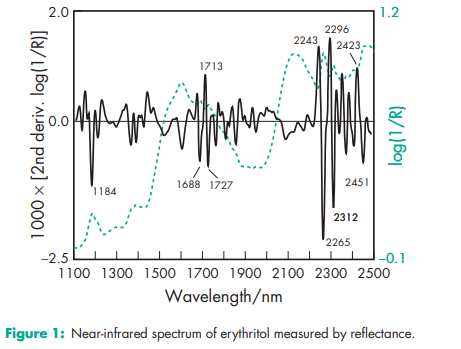
Acidity/alkalinity pH = 5–7 at 258C for a 5% w/v aqueous solution. Boiling point 329–3318C Caloric value 0.8 kJ/g Density 1.45 g/cm3 Dissociation constant pKa = 13.90 at 188C Heat of solution 22 kJ/mol Hygroscopicity Erythritol is nonhygroscopic; it absorbs approximately 1% w/w of water at 95% relative humidity (RH). Melting point 121.58C, with decomposition at 1608C. NIR spectra see Figure 1. Solubility Soluble 1 in 3 of water; slightly soluble in ethanol (95%); practically insoluble in ether and fats. Viscosity (dynamic) 3 mPa s (3 cP) at 608C for a 30% w/w solution.
Erythritol has very good thermal and chemical stability. It is nonhygroscopic, and at 258C does not significantly absorb additional water up to a relative humidity (RH) of more than 80%. Erythritol resists decomposition both in acidic and alkaline media and remains stable for prolonged periods at pH 2–10.(10) When stored for up to 4 years in ambient conditions (208C, 50% RH) erythritol has been shown to be stable.
Erythritol is incompatible with strong oxidizing agents and strong bases.
Erythritol is a starch-derived product. The starch is enzymatically hydrolyzed into glucose which is turned into erythritol via a fermentation process, using osmophilic yeasts or fungi (e.g. Moniliella pollinis, or Trichosporonoides megachiliensis).
Erythritol is used in oral pharmaceutical formulations, confectionery, and food products. It is generally regarded as a nontoxic, nonallergenic, and nonirritant material.(12) However, there has been a case report of urticaria caused by erythritol.(13) The low molecular weight of erythritol allows more than 90% of the ingested molecules to be rapidly absorbed from the small intestine;(14) it is not metabolized and is excreted unchanged in the urine. Erythritol has a low caloric value (0.8 kJ/g). The WHO has set an acceptable daily intake of ‘not specified’ for erythritol.(12) Erythritol is noncariogenic; preliminary studies suggest that it may inhibit the formation of dental plaque.(15) In general, erythritol is well-tolerated;(16–18) furthermore, excessive consumption does not cause laxative effects. There is no significant increase in the blood glucose level after oral intake, and glycemic response is very low, making erythritol suitable for diabetics. LD50 (mouse, IP): 8–9 g/kg(12) LD50 (rat, IV): 6.6 g/kg LD50 (rat, oral): >13 g/kg
Observe normal precautions appropriate to the circumstances and quantity of the material handled. Eye protection and a dust mask or respirator are recommended.
GRAS listed. Accepted for use as a food additive in Europe
Mannitol; sorbitol; xylitol.
Active ingredients can be granulated with erythritol and binders such as maltodextrin or carboxymethylcellulose, resulting in coarser granules with improved flowability.(5) Coprocessing erythritol with a small amount of maltodextrin results in a proprietary compound that may be used in direct compression.(19) A specification for erythritol is included in the Japanese Pharmaceutical Excipients (JPE).(20) The EINECS number for erythritol is 205-737-3. The PubChem Compound ID (CID) for erythritol is 8998