

Name: Ethyl Acetate
CAS No: Ethyl acetate [141-78-6]
BP: Ethyl Acetate PhEur: Ethyl Acetate USP-NF: Ethyl Acetate
Acetic acid ethyl ester; acetic ester; acetic ether; acetoxyethane; aethylis acetas; aethylium aceticum; ethyl ethanoate; ethylis acetas; vinegar naphtha
Ethyl acetate [141-78-6]
C4H8O2 88.1
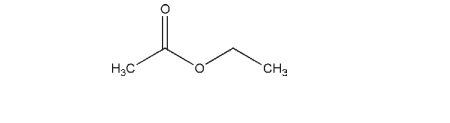
In pharmaceutical preparations, ethyl acetate is primarily used as a solvent, although it has also been used as a flavoring agent. As a solvent, it is included in topical solutions and gels, and in edible printing inks used for tablets. Ethyl acetate has also been shown to increase the solubility of chlortalidone(1) and to modify the polymorphic crystal forms obtained for piroxicam pivalate,(2) mefenamic acid,(3) and fluconazole,(4) and has been used in the formulation of microspheres.(5–8) Ethyl acetate has been used as a solvent in the preparation of a liposomal amphotericin B dry powder inhaler formulation.(9) Its use as a chemical enhancer for the transdermal iontophoresis of insulin has been investigated.(10) In food applications, ethyl acetate is mainly used as a flavoring agent. It is also used in artificial fruit essence and as an extraction solvent in food processing.
Ethyl acetate is a clear, colorless, volatile liquid with a pleasant fruity, fragrant, and slightly acetous odor, and has a pleasant taste when diluted. Ethyl acetate is flammable.
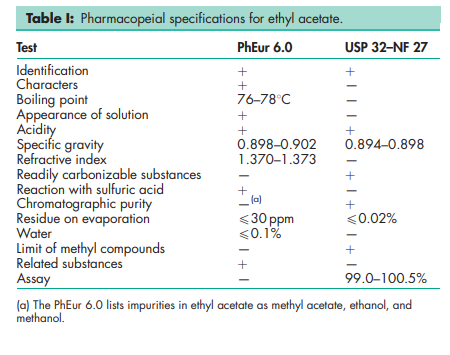
See Table I.
Autoignition temperature 486.18C Boiling point 778C Dielectric constant 6.11 Density 0.902 g/cm3 at 208C Explosive limit 2.2–11.5% (volume in air) Flash point þ7.28C (open cup); 5.08C (closed cup). Freezing point 83.68C NIR spectra see Figure 1. Partition coefficient log P (octanol/water) = 0.7 Refractive index n D 20 = 1.3719 Solubility Soluble 1 in 10 of water at 258C; ethyl acetate is more soluble in water at lower temperatures than at higher temperatures. Miscible with acetone, chloroform, dichloromethane, ethanol (95%), and ether, and with most other organic liquids. Vapor density 3.04 (air = 1)
Ethyl acetate should be stored in an airtight container, protected from light and at a temperature not exceeding 308C. Ethyl acetate is slowly decomposed by moisture and becomes acidic; the material can absorb up to 3.3% w/w water. Ethyl acetate decomposes on heating to produce ethanol and acetic acid, and will emit acrid smoke and irritating fumes. It is flammable and its vapor may travel a considerable distance to an ignition source and cause a ‘flashback’. The alkaline hydrolysis of ethyl acetate has been shown to be inhibited by polyethylene glycol and by mixed micelle systems.(1
Ethyl acetate can react vigorously with strong oxidizers, strong alkalis, strong acids, and nitrates to cause fires or explosions. It also reacts vigorously with chlorosulfonic acid, lithium aluminum hydride, 2-chloromethylfuran, and potassium tert-butoxide.
Ethyl acetate can be manufactured by the slow distillation of a mixture of ethanol and acetic acid in the presence of concentrated sulfuric acid. It has also been prepared from ethylene using an aluminum alkoxide catalyst.
Ethyl acetate is used in foods, and oral and topical pharmaceutical formulations. It is generally regarded as a relatively nontoxic and nonirritant material when used as an excipient. However, ethyl acetate may be irritant to mucous membranes, and high concentrations may cause central nervous system depression. Potential symptoms of overexposure include irritation of the eyes, nose, and throat, narcosis, and dermatitis. Ethyl acetate has not been shown to be a human carcinogen or a reproductive or developmental toxin. The WHO has set an estimated acceptable daily intake of ethyl acetate at up to 25 mg/kg body-weight.(12) In the UK, it has been recommended that ethyl acetate be temporarily permitted for use as a solvent in food and that the maximum concentration consumed in food should be set at 1000 ppm.(13) LD50 (cat, SC): 3.00 g/kg(14) LD50 (guinea-pig, oral): 5.50 g/kg LD50 (guinea-pig, SC): 3.00 g/kg LD50 (mouse, IP): 0.709 g/kg LD50 (mouse, oral): 4.10 g/kg LD50 (rabbit, oral): 4.935 g/kg LD50 (rat, oral): 5.62 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended. In the UK, the workplace exposure limit for ethyl acetate is 400 ppm (short-term) and 200 ppm (long-term).(15)
Included in the FDA Inactive Ingredients Database (oral tablets and sustained-action tablets; topical and transdermal preparations). Included in nonparenteral medicines licensed in the UK (tablets, topical solutions, and gels). Ethyl acetate is also accepted for use in food applications in a number of countries including the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients
—
The following azeotropic mixtures have been reported: Ethyl acetate (93.9% w/w)–water (6.1% w/w), boiling point 70.48C Ethyl acetate (83.2% w/w)–water (7.8% w/w)–ethanol (9.0% w/w), boiling point 70.38C Ethyl acetate (69.4%)–ethanol (30.6%), boiling point 71.88C Ethyl acetate (77%)–propan-2-ol (23%), boiling point 74.88C A specification for ethyl acetate is contained in the Food Chemicals Codex (FCC).(16) The EINECS number for ethyl acetate is 205-500-4. The PubChem Compound ID (CID) for ethyl acetate is 8857.