

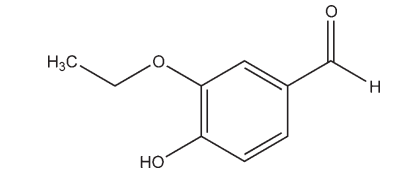
Name: Ethyl Vanillin
CAS No: 3-Ethoxy-4-hydroxybenzaldehyde [121-32-4]
USP-NF: Ethyl Vanillin
Bourbonal; ethylprotal; ethylprotocatechuic aldehyde; 4-hydroxy3-ethoxybenzaldehyde; Rhodiarome; vanillal.
3-Ethoxy-4-hydroxybenzaldehyde [121-32-4]
C9H10O3 ,166.18
Ethyl vanillin is used as an alternative to vanillin, i.e. as a flavoring agent in foods, beverages, confectionery, and pharmaceuticals. It is also used in perfumery. Ethyl vanillin possesses a flavor and odor approximately three times as intense as vanillin; hence the quantity of material necessary to produce an equivalent vanilla flavor may be reduced, causing less discoloration to a formulation and potential savings in material costs. However, exceeding certain concentration limits may impart an unpleasant, slightly bitter taste to a product due to the intensity of the ethyl vanillin flavor. See Table I.

White or slightly yellowish crystals with a characteristic intense vanilla odor and flavor.
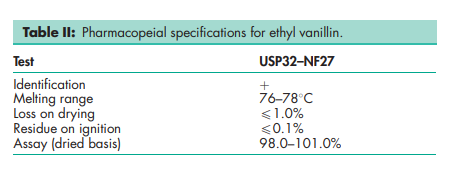
See Table II
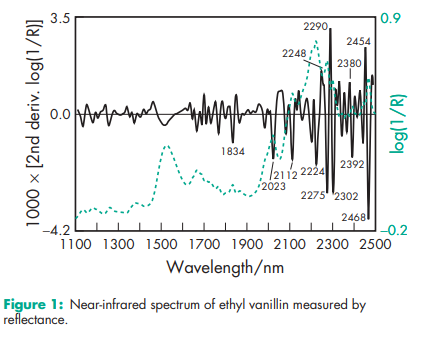
Boiling point 2858C Density (bulk) 1.05 g/cm3 Flash point 1278C Melting point 76–788C NIR spectra see Figure 1. Solubility see Table III.

Store in a well-closed container, protected from light, in a cool, dry place. See Vanillin for further information.
Ethyl vanillin is unstable in contact with iron or steel, forming a redcolored, flavorless compound. In aqueous media with neomycin sulfate or succinylsulfathiazole, tablets of ethyl vanillin produced a yellow color.
Unlike vanillin, ethyl vanillin does not occur naturally. It may be prepared synthetically by the same methods as vanillin, using guethol instead of guaiacol as a starting material
Ethyl vanillin is generally regarded as an essentially nontoxic and nonirritant material. However, cross-sensitization with other structurally similar molecules may occur; see Vanillin. The WHO has allocated an acceptable daily intake for ethyl vanillin of up to 3 mg/kg body-weight.(2) LD50 (guinea pig, IP): 1.14 g/kg(3,4) LD50 (mouse, IP): 0.75 g/kg LD50 (rabbit, oral): 3 g/kg LD50 (rabbit, SC): 2.5 g/kg LD50 (rat, oral): 1.59 g/kg LD50 (rat, SC): 3.5–4.0 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection is recommended. Heavy airborne concentrations of dust may present an explosion hazard.
GRAS listed. Included in the FDA Inactive Ingredients Database (oral capsules, suspensions, and syrups). Included in nonparenteral medicines licensed in the UK.
Vanillin.
Ethyl vanillin can be distinguished analytically from vanillin by the yellow color developed in the presence of concentrated sulfuric acid. The EINECS number for ethyl vanillin is 204-464-7. The PubChem Compound ID (CID) for ethyl vanillin is 8467.