

Name: Ethylene Vinyl Acetate
CAS No: Ethylene vinyl acetate copolymer [24937-78-8]
None adopted.
Acetic acid, ethylene ester polymer with ethane; CoTran; ethylene/ vinyl acetate copolymer; EVA; EVA copolymer; EVM; poly(ethylene-co-vinyl acetate); VA/ethylene copolymer; vinyl acetate/ethylene copolymer.
Ethylene vinyl acetate copolymer [24937-78-8]
(CH2CH2)x[CH2CH(CO2CH3)]y
Ethylene vinyl acetate copolymer is a random copolymer of ethylene and vinyl acetate.
Ethylene vinyl acetate copolymers are used as membranes and backings in laminated transdermal drug delivery systems. They can also be incorporated as components in backings in transdermal systems. Ethylene vinyl acetate copolymers have been shown to be an effective matrix and membrane for the controlled delivery of atenolol(1,2) triprolidine,(3,4) and furosemide.(5) The system for the controlled release of atenolol can be further developed using ethylene vinyl acetate copolymers and plasticizers.(1)
Ethylene vinyl acetate is available as white waxy solids in pellet or powder form. Films are translucent.
—
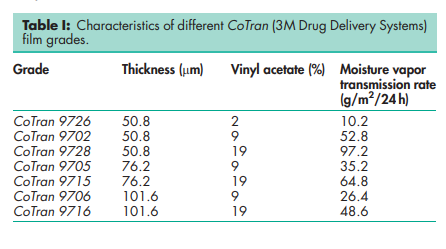
Density 0.92–0.94 g/cm3 Flash point 2608C Melting point 75–1028C depending on polymer ratios. Moisture vapor transmission rate see Table I. Thickness see Table I. Vinyl acetate content see Table I.
Ethylene vinyl acetate copolymers are stable under normal conditions and should be stored in a cool, dry place. Films of ethylene vinyl acetate copolymers should be stored at 0–308C and less than 75% relative humidity.
Ethylene vinyl acetate is incompatible with strong oxidizing agents and bases.
Various molecular weights of random ethylene vinyl acetate copolymers can be obtained by high-pressure radical polymerization, bulk continuous polymerization, or solution polymerization.
Ethylene vinyl acetate is mainly used in topical pharmaceutical applications as a membrane or film backing. Generally it is regarded as a relatively nontoxic and nonirritant excipient.
Observe normal precautions appropriate to the circumstances and quantity of material handled. Ethylene vinyl acetate powder may form an explosive mixture with air.
Included in the FDA Inactive Ingredients Database (intrauterine suppository; ophthalmic preparations; periodontal film; transdermal film). Included in nonparenteral medicines licensed in the UK.
—
Ethylene vinyl acetate copolymers have a wide variety of industrial uses. Properties of ethylene vinyl acetate copolymer films in terms of oxygen and moisture transfer rate are related to the vinyl acetate content and thickness. Higher levels of vinyl acetate result in increased lipophilicity, increased oxygen and moisture vapor permeability, and increased clarity, flexibility, toughness, and solvent solubility. The PubChem Compound ID (CID) for ethylene vinyl acetate is 32742