

Name: Fumaric Acid
CAS No: (E)-2-Butenedioic acid [110-17-8]
USP-NF: Fumaric Acid
Allomaleic acid; allomalenic acid; boletic acid; butenedioic acid; E297; 1,2-ethenedicarboxylic acid; lichenic acid; trans-butenedioic acid; NSC-2752; trans-1,2-ethylenedicarboxylic acid; U-1149; USAF EK-P-583.
(E)-2-Butenedioic acid [110-17-8]
C4H4O4 , 116.07
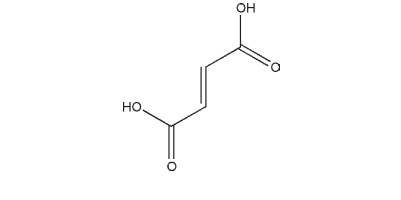
Fumaric acid is used primarily in liquid pharmaceutical preparations as an acidulant and flavoring agent. Fumaric acid may be included as the acid part of effervescent tablet formulations, although this use is limited as the compound has an extremely low solubility in water. It is also used as a chelating agent which exhibits synergism when used in combination with other true antioxidants. In the design of novel pelletized formulations manufactured by extrusion–spheronization, fumaric acid was used to aid spheronization, favoring the production of fine pellets.(1) It has also been investigated as an alternative filler to lactose in pellets.(2) Fumaric acid has been investigated as a lubricant for effervescent tablets,(3) and copolymers of fumaric acid and sebacic acid have been investigated as bioadhesive microspheres.(4) It has been used in film-coated pellet formulations as an acidifying agent and also to increase drug solubility.(5) Fumaric acid is also used as a food additive at concentrations up to 3600 ppm, and as a therapeutic agent in the treatment of psoriasis and other skin disorders.(6)
Fumaric acid occurs as white, odorless or nearly odorless, granules or as a crystalline powder that is virtually nonhygroscopic.
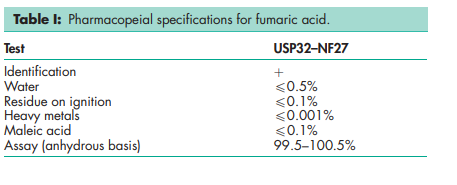
See Table I.
Acidity/alkalinity pH = 2.45 (saturated aqueous solution at 208C); pH = 2.58 (0.1% w/v aqueous solution at 258C); pH = 2.25 (0.3% w/v aqueous solution at 258C); pH = 2.15 (0.5% w/v aqueous solution at 258C). Boiling point 2908C (sealed tube) Density 1.635 g/cm3 at 208C Density (bulk) 0.77 g/cm3 Density (tapped) 0.93 g/cm3 Dissociation constant pKa1 = 3.03 at 258C; pKa2 = 4.54 at 258C. Melting point 2878C (closed capillary, rapid heating); partial carbonization and formation of maleic anhydride occur at 2308C (open vessel); sublimes at 2008C. NIR spectra see Figure 1. Solubility see Table II.

Fumaric acid is stable although it is subject to degradation by both aerobic and anaerobic microorganisms. When heated in sealed vessels with water at 150–1708C it forms DL-malic acid. The bulk material should be stored in a well-closed container in a cool, dry place.
Fumaric acid undergoes reactions typical of an organic acid.
Commercially, fumaric acid may be prepared from glucose by the action of fungi such as Rhizopus nigricans, as a by-product in the manufacture of maleic and phthalic anhydrides, and by the isomerization of maleic acid using heat or a catalyst. On the laboratory scale, fumaric acid can be prepared by the oxidation of furfural with sodium chlorate in the presence of vanadium pentoxide.
Fumaric acid is used in oral pharmaceutical formulations and food products, and is generally regarded as a relatively nontoxic and nonirritant material. However, acute renal failure and other adverse reactions have occurred following the topical and systemic therapeutic use of fumaric acid and fumaric acid derivatives in the treatment of psoriasis or other skin disorders.(6,7) Other adverse effects of oral therapy have included disturbances of liver function, gastrointestinal effects, and flushing.(6) The WHO has stated that the establishment of an estimated acceptable daily intake of fumaric acid or its salts was unnecessary since it is a normal constituent of body tissues.(8) LD50 (mouse, IP): 0.1 g/kg(9) LD50 (rat, oral): 9.3 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Fumaric acid may be irritating to the skin, eyes, and respiratory system, and should be handled in a wellventilated environment. Gloves and eye protection are recommended.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (oral capsules, suspensions, syrups, extended release and sustained action chewable tablets). Included in the Canadian List of Acceptable Nonmedicinal Ingredients.
Citric acid monohydrate; malic acid; tartaric acid.
A specification for fumaric acid is contained in the Food Chemical Codex (FCC).(10) The EINECS number for fumaric acid is 203-743-0. The PubChem Compound ID (CID) for fumaric acid is 444972.